Plastics are produced in large quantities due to the high demand for their use in agriculture, households, automobiles, packing materials, toys, electronics, and a variety of other applications. The demand for plastics has increased by 5% every year since 1990. The increase in plastic use correspondingly increases the number of plastic wastes being produced. Plastic waste can be classified into at least two categories: municipal and industrial. Industrial plastics are generally more homogeneous and contamination-free, making them useful for downcycling into lower-grade plastic products. Municipal plastics tend to be more heterogeneous and contain extraneous materials.
In general, about 10.675.1 wt% of municipal wastes consists of plastics. The municipal plastic waste consists primarily of low-density polyethylene (LDPE), high-density polyethylene (HDPE), poly(ethylene terephthalate) (PET), polypropylene (PP), polystyrene (PS), and poly(vinyl chloride) (PVC). Overall, about 50-70% of the total plastic waste is packaging materials derived from polyethylene, polypropylene, polystyrene, and polyvinyl chloride. On average, polyethylene makes up the greatest fraction of all plastic wastes (69%), especially plastic bags, and polyethylene comprises 63% of the total packaging waste.
Polyethylene (PE, low density, and high density) and polypropylene (PP) are the most widely used plastics. HDPE is recyclable and can be found in plastic bottles, storage boxes, pipes and cable insulations, and many other uses. LDPE can be utilized for making computer parts, toys, soft bottles, wrappers, back sheets for diapers, and numerous additional applications. In the packaging industry, all three polymers, PP, PS, and PE, are widely used. A copolymer of ethylene and propylene is also frequently employed, e.g., as rubber and in computers. Poly (vinyl chloride) (PVC) is another popular plastic that is associated with a variety of applications, e.g., plumbing pipes, electrical cable insulation, tubings, automobile seat covers, and rubber replacement in some applications. Poly(ethylene terephthalate) (PET) is also a common polymer that has found many applications, e.g., films, fibers, food containers, and beverage bottles.
Plastic disposal is a major concern in many countries, including the United States. After its initial use, over 60% of the total plastic solid waste (PSW) produced is discarded in landfills throughout the world. Less than 10% of plastic waste is recycled. This is problematic, as plastic waste is a major environmental threat due to its non-degradability, its potential health risks to aquatic and terrestrial animals, and its impact on environmental pollution. The waste plastics ending up in the ocean make a huge plastic soup and garbage patch like the great Pacific garbage patch risking the health of aquatic animals. In the last 40 years, the plastic waste in the North Pacific Ocean has increased by 100-fold. The plastic footprint is considered more dangerous than the carbon footprint. Some of the proposed solutions for PSW management are incineration or mechanical recycling. However, incineration can contribute to pollution by causing harmful and toxic emissions. Additionally, both of these processes are costly and may or may not be economically viable in different situations.
Aside from the challenge of plastic waste disposal, another global issue is the energy crisis. Transportation consumes one-third of the world’s energy. The main energy sources for transportation are fossil fuels, coal, oil, and natural gas, all of which are non-renewable sources of energy. Fossil fuels are also major sources of environmental pollution, greenhouse gases, and ocean acidification. Today, these fuels are being consumed at an unsustainably high rate throughout the world. Even though more than a hundred billion tons of oil and gas have been discovered in the last 40 years, the rate at which it is consumed has also increased. The US alone consumes one-quarter of the global oil supply, while only having 1.6% of the total oil reserve. At the current rate of consumption, the global supply of fossil fuels will be depleted within 40–70 years. Among 195 countries in the world, only 40 can produce fossil fuel, and for some countries, their independent oil reserves still do not satisfy all their energy The challenges of plastic waste management and increasing energy demand can simultaneously be addressed by the production of fuel from plastics, and a number of research groups are currently developing this capacity. The fuels produced from plastics can be clean and have fuel properties similar to fossil fuels. HDPE, LDPE, and PP (plastics identification codes 2, 4 and 5, respectively) are polymers containing only carbon and hydrogen.
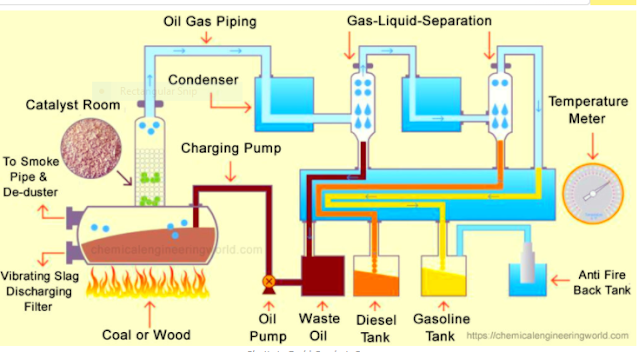
Unlike biofuels, the absence of oxygen and higher carbon and hydrogen content (Table 1) in plastic fuels avoids the need for further upgrading. The absence of water in plastic fuels makes the calorific value very high, and the absence of oxygen content makes the fuel non-acidic and non-corrosive, unlike biofuel. Therefore, conversion of these plastic wastes to usable oil is a growing and important field of study that can potentially mitigate the energy crisis. However, the techno-economic evaluation plays an important role in the commercial success of the plastics-to-fuel conversion. The thermal and catalytic processes of converting waste plactics into fuels are promising techniques to eliminate the refuse which otherwise is harmful to the enivironment and decrease the dependence on fossil fuels. Thermal degradation decomposes plastic into three fractions: gas, crude oil, and solid residue.
Crude oil from non-catalytic pyrolysis is usually composed of higher boiling point hydrocarbons. The optimization of conversion parameters such as the choice of catalyst, reactor design, pyrolysis temperature, and plastic-to-catalyst ratio plays a very important role in the efficient generation of gasoline and diesel-grade fuel. The use of a catalyst for thermal conversion lowers the energy required for conversion, and the catalyst choice is important for efficient fuel production. The suitable selection of catalysts can increase the yield of crude oil with lower hydrocarbon content. Co-pyrolysis of plastics with coal or shale oil improves crude oil quality by decreasing its viscosity. A large number of publications have appeared on various processes, and continued improvements and/or innovations are expected in the future. Further investigations on the catalytic systems are required in order to advance the field, particularly to enhance the added value of fuels and to minimize the use of energy. This review aims to provide both the highlights of the remarkable achievements of this field and the milestones that need to be achieved in the future.
Conclusions
Fuel production from plastics is attractive because it simultaneously addresses the issues of waste management and alternative energy generation. The optimization of conversion parameters such as the choice of catalysts, reactor design, pyrolysis temperature, and plastic-to-catalyst ratio plays a very important role in the efficient processing of gasoline and diesel-grade fuel. The use of a catalyst for thermal conversion lowers the energy required for conversion, and catalyst choice is important for efficient fuel production. A catalyst with higher acidic properties and a higher porosity can enhance conversion; for example, FCC catalysts catalyze the production of aromatic and gaseous hydrocarbons, whereas dual catalysts like zeolite and metal-loaded catalysts can be used for producing hydrocarbons and isomerized products, which increase the octane rating of fuels. Uniform distribution of heat and mass is another important factor in conversion.
A fluidized bed reactor is an excellent choice to ensure uniform distribution of heat, resulting in better conversion. Despite the recent advancements in plastic conversion technology, several issues remain. A major challenge with the production of fuel from plastic solid waste is the presence of PVC, which produces HCl gas during pyrolysis. However, researchers have been able to remove chlorine to some extent by using HCl adsorbents to pretreat the plastics. Catalysts will also need improvement. For example, it will be useful to use dual catalysts with a combination of high acidic properties, porosity, and hydrogenation properties, identify accessible and cheaper catalysts to use on large scales and prevent deactivation and increase catalyst reuse. In fact, the use of metal-loaded biochar may partly mitigate the high cost of catalysts. It is very important to devise efficient methods to produce fuel from mixed plastics or to exclude or remove PET and PVC from mixed plastics before pyrolysis.
The product yield and quality can be improved by finding alternative heating modes such as IR and microwave heating to reach the final temperature quickly. A thorough study of the effect of heating rates on product yields and distribution will be very important to produce fuel-range hydrocarbons. There are many other factors that affect the economics of fuel production in terms of raw material handling, such as collection, classification, washing and shredding, transportation, electric energy, heating gas, and cooling water. The cost of each step varies from the U.S. $65 to $400/ton. The landfill costs in USA may vary from $25 to $150/ton depending on the region of the country. It was reported that a well-run curbside recycling program can cost, under general circumstances, between $50 and $150/ton. Therefore, the cost of recycling plastic is relatively inexpensive compared to the production of fuel from plastic, and the cost of fossil fuel is currently cheaper than the production of fuel from plastics. However, the demand for fossil fuel, the issue of disposing of plastic waste, and the environmental effect make the production of fuel from plastic economically attractive in the longer term. Certainly continued research and development is needed in this field